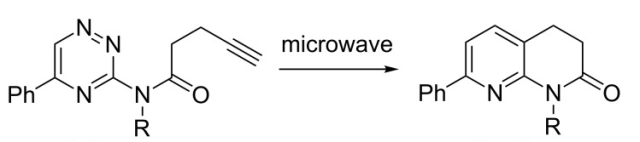
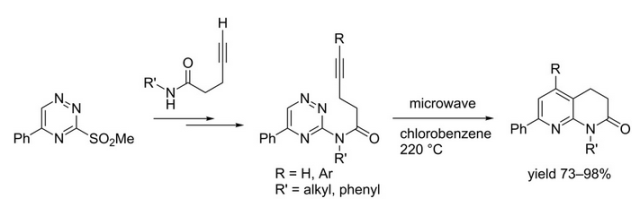
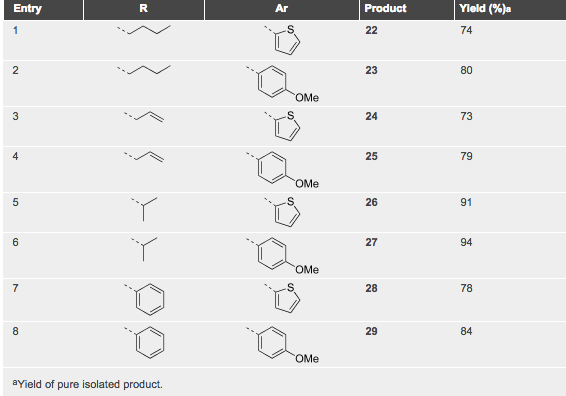
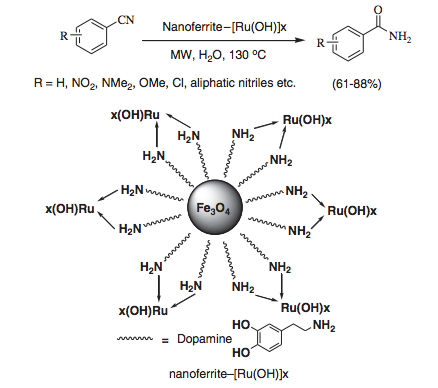
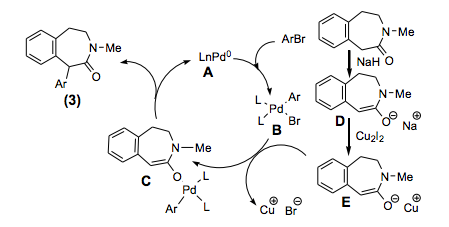
The field of cycloadditions under microwave control is vast and has been represented in a fair number of books and review articles. What is noteworthy is the array of important advanced intermediates that can be used as handles or core heterocycles and complex carbocyles for future chemistry. Without a lecture I will highlight a few examples from a review article from Erik Van der Eycken (a well known microwave strategists) in Chem Soc Reviews 2010 (can be found as a pdf with a quick google search).
The review is broken down into the types of cycloadditions starting with the familiar [3+2] triazole forming reactions (click, click) then proceeds to other [3+2], [4+2] and ends with [2+2] reactions. At the risk of labeling these tranformations as percyclic reactions, we will hand wave and and end that some are very quick step transformations (and often concerted) and their selectivity patterns are well documented. I certainly have a fondness for these reactions with my background in isoxazole, diels-alder and isomunchnone cascade studies — so I have to like these, right? Well below, I have used a couple of examples from the review to give you a flavor, but left the triazole chemistry for you:
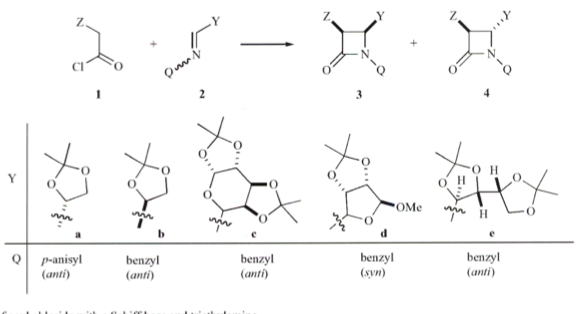
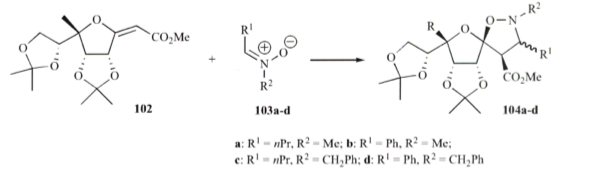
[3+2] Polycyclic pyrrolidines 1,3-dipolar process
Condensation of diaminoarene to form an N-allylhydrobendimidazole intermediate provided the requisite backbone to react with a variety of amino esters. The s-ylide could be then be react with the 1,3-dipole under microwave heating in toluene at 130C for 20-30 minutes to provide the fused system in high yields – much faster, cleaner and higher yields when compared with conventional heating.

Isoxazole formation via nitrile oxide intermediate
This was a rather slick method when you can use it. The group started with an aldoxime — generated the corresponding nitrile oxide alumina-NCS under microwave heating at 110-150C with a 3 min pulse of microwave power and the advanced acetylene was added for a 2 step-one pot solid phase formation of a library of isoxazoles — sure beats the classical ways of the formation of nitrile oxides and when you can get a terminal alkyne — take it. It is mentioned that the yields were much higher and less time consuming than the traditional solution phase heating of up to 5 hours.

[4+2] Pendant furan cycloaddition-rearrangement a plus cascade reaction to Strychnine
Al Padwa seemed to always have an elegant approach to complex structures – I was lucky enough to have a number of interactions with him and a number of his students who had passed through Emory University (of course I got the smelly Pummerer reactions, LOL!). Rather than looking into his volumes of straight-up DA reactions we will go to a COOL example. The example highlighted involved the use of a pendant amido-furan under a Mg-I2 catalysed [4+2] cycloaddition reaction followed by rearrangement to form an advanced tetracycle on the way to Strychnine (this is a great example for anyone drawing the arrows to push the formation of the intermediate cycloadduct then rearrangement — I think I still have it on a napkin someplace).

And lastly because the formation of 4,5,6 and 7 positions of an indole can be difficult to build substitution patterns the use of cycloaddition strategies can prove highly useful. The [4+2] cycloaddition of a representative set of 2H-pyran-2-ones and a suitable alkyne to form the requisite amido-arene with the double bond positioned for ring closure to the indole system, which was also performed under microwave heating. Although this group used a two-step 2-pot approach, it is likely that conditions could be used to add reagents for this transformation to be done in one-pot — I know the process chemists would find a way. Great example of the use of this strategy.

Many more examples are available to read — and too many for a single post so enjoy the article.